


The volume of container 1 is 1.30 L, and it contains 6.70 mol of the gas. Gaseous chlorine is held in two separate containers at identical temperature and pressure.Determine the temperature at which the volume of the gas is 3.42 L. The gas occupies 3.62 L of volume when the temperature is 21.6☌.
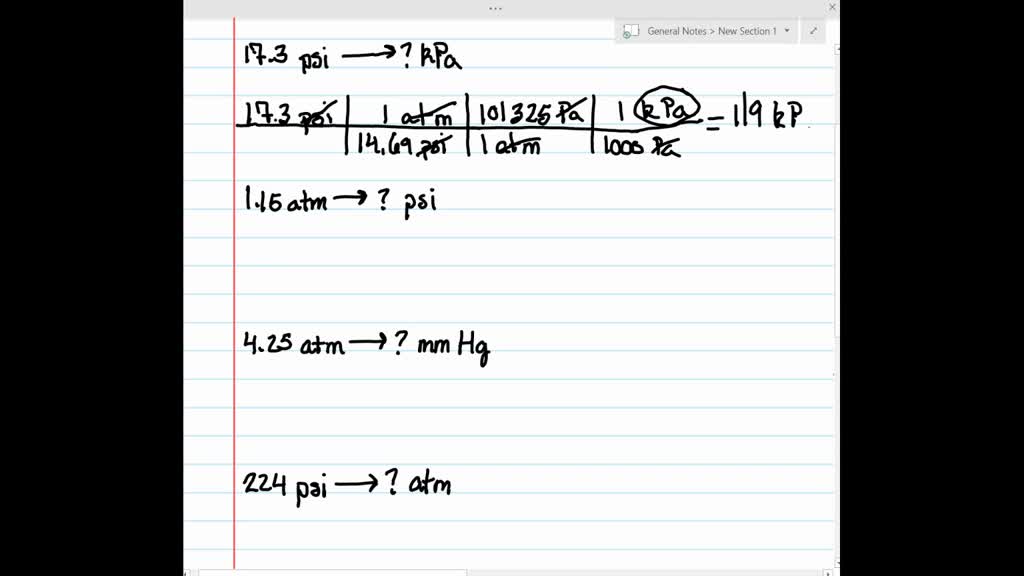
#755 mmhg to atm download
To download a copy of the assignment, please click on the link Sample Questions.Īs you work these matter and measurement problems, consider and explain:
